Be Cautious and Avoid Using Medication from Series A00101: Inspection Body
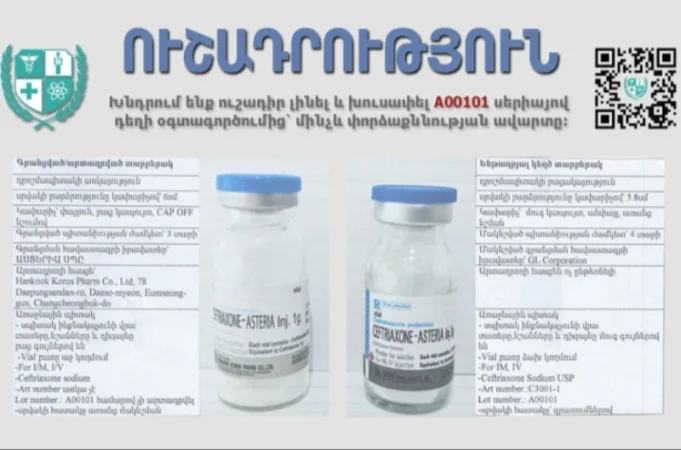
On March 5, 2024, according to the letter No. 01/1148-24 received from the Expert Center for Medications and Medical Technologies named after Academician Emil Gabrielian of the Ministry of Health of the Republic of Armenia, and the attached conclusion No. 01CA24, it has been determined that the medication Ceftriaxone-Asteria Inj. 1g powder for solution for injection, produced by Hancook Corus Pharm Co., Ltd remains unexamined according to the procedures established by law, and there is a lack of relevant data regarding its legal import into Armenia according to the database of the Expert Center.
Meanwhile, it is reported that there is suspicion of the mentioned series of the medication being counterfeit, and the circumstances are being clarified.
Dear citizens, since there is a suspicion of a first-class defect that may require a recall, we kindly ask you to be cautious and avoid using the medication from series A00101, as it may be counterfeit, until the examination is completed.
Additionally, some differences between the registered/produced and the alleged counterfeit version are presented.







